Indicated CKD with no Diagnosis Report
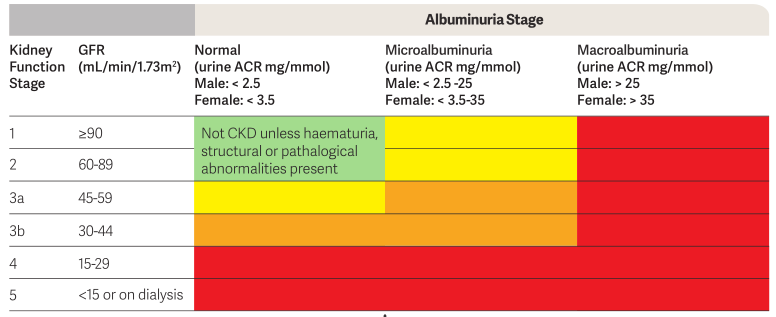
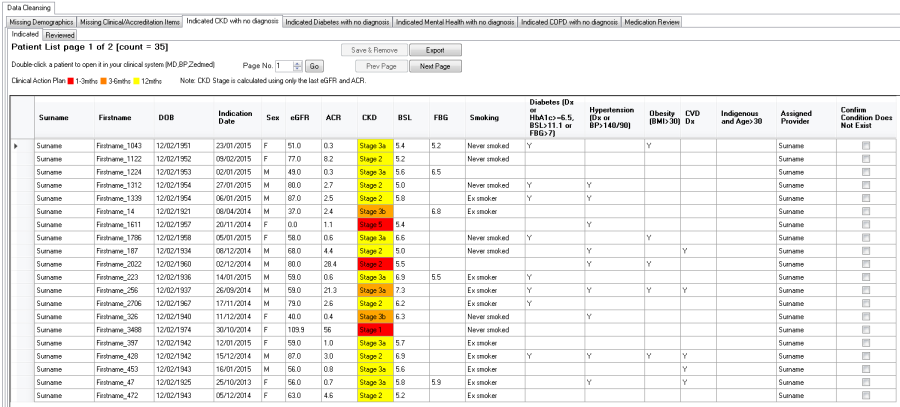
The "Indicated CKD with no Diagnosis" Report displays patients where the staging of CKD, as determined by the combined results of kidney function (eGFR) and kidney damage (the level of albuminuria using ACR), indicates the possibility of CKD.
The report displays:
- eGFR, ACR and the resulting Indicated Staging of CKD
- Color code for the Clinical Action Plan to manage CKD
- Other CKD Risk Factors
Patients on dialysis are displayed as 'level 5' CKD.
Patients with an eGFR = 0 and no ACR are displayed without action plan level and just with the word 'Stage' to indicate that there is some data in their record that requires review.
Red | Orange | Yellow | No Colour |
For more information refer to Kidney Health Australia web site and the CKD Guideline Booklet available at www.kidney.org.au
Detection of CKD requires follow up testing once an abnormal result is recorded. The outcome of 3 tests over a 3 month period will inform a diagnosis. CAT4 is only using the most recent eGFR and ACR, not the last 3 results. This will
•inform a diagnosis if 3 tests have been done or
•flag that a patient should be recalled for follow up testing if there are not 3 results in the Patient Record.
Pen CS wishes to acknowledge the input of Dr Craig Nelson in the creation of this report.
Indicated Diabetes with no Diagnosis Report
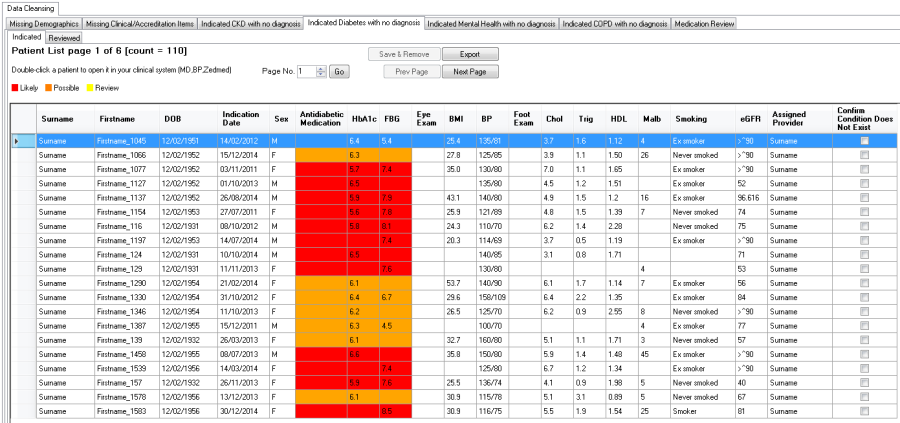
The "Indicated Diabetes with no Diagnosis" Report will display the likelihood of Diabetes based on HbA1c, Anti-diabetic Medication and/or FBG being recorded in the patient record without a diagnosis:
•Red = likely
•Orange = possible
•Yellow = flag for review
Red: ANY of the following | Orange: ANY of the following | Yellow : ANY of the following |
Medications need to be long term to be included.
The report displays
•HbA1c, Anti-diabetic Medication, FBG and the resulting likelihood
•Diabetes Care items that have been recorded
Pen CS wishes to acknowledge the input of Dr Ralph Audehm in the creation of this report.
The HbA1c test is used to monitor Diabetic patients so the fact that there is an HbA1c in the patient's record indicates they could be diabetic. The HbA1c test can also be used by clinicians for pre-diabetics or patients with impaired glucose test. So it is just alerting the practice to review. The report shows other data items such as eye check and foot check. These are presented to show other activities that are being carried out that are part of the SIP cycle for reference.
Indicated Mental Health with no Diagnosis Report
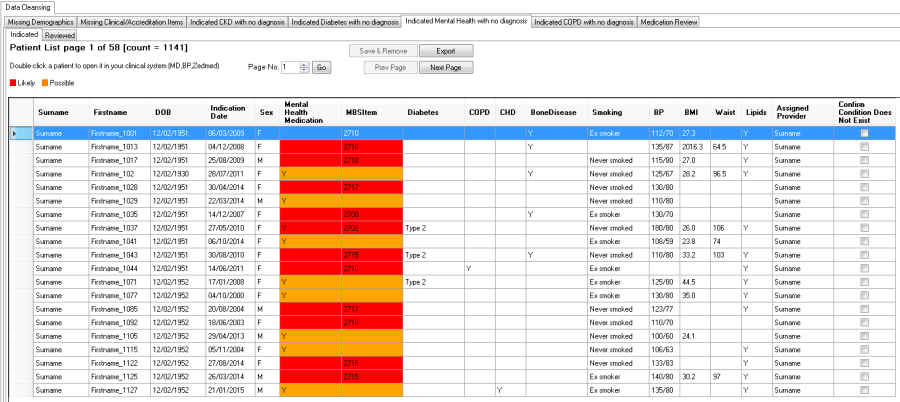
The "Indicated Mental Health with no diagnosis" Report will display the likelihood of a Mental Health condition based on a mental health medication or a mental health care plan being recorded in the patient record without a diagnosis. Any mental health diagnoses included in the existing CAT4 reports/filters is considered regardless if the diagnosis is marked as active or inactive in the patient record.
•Red = likely
•Orange = possible
RED: | ORANGE: |
The report displays
•MH medication, MH MBS item numbers and the resulting likelihood
•Chronic disease co-morbidities
•Other MH risk factors
Pen CS wishes to acknowledge the input of Noel Stewart from NEVDGP and his work in the use of CAT to manage patients with chronic disease and mental health conditions in the creation of this report.
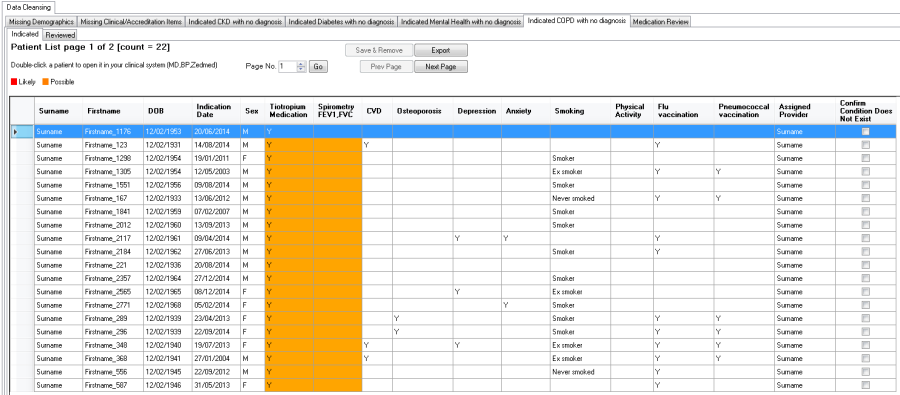
Indicated COPD with no Diagnosis Report
The "Indicated COPD with no diagnosis" Report will display the likelihood of COPD based on relevant respiratory medication or an adverse spirometry reading being recorded in the patient record without a diagnosis:
•Red = likely
•Orange = possible
Please note that currently only POST values for spirometry are taken into consideration in this report!
RED: | ORANGE: |
The report displays
•COPD medication, spirometry readings and the resulting likelihood
•Chronic disease co-morbidities
•Management items
For more information refer to
- 'The Spirometry Handbook' available from http://www.nationalasthma.org.au
- 'The Pocket Guide to COPD Diagnosis, Management, and Prevention' available from http://www.goldcopd.org
- Respiratory medication filter in CAT4: Respiratory Medications
COPD Management Tiotropium (LAMA) | Generic Name: Tiotropium |
COPD Management - Other (LAMA) | Trade Name: BRETARIS GENUAIR (ACLIDINIUM) SEEBRI BREEZHALER (GLYCOPYRRONIUM) INCRUSE ELLIPTA (UMECLIDINIUM) |
COPD LABA Medications | ONBREZ BREEZHALER (INDACATEROL) |
COPD LAMA/LABA Medications | SPILOTO RESPIMAT (Tiotropium/Olodatererol) BRIMICA GENUAIR (Aclidinium/Formoterol) ULTIBRO BREEZHALER (Indacaterl/glycopyrroium) ANORO ELLIPTA (Umeclinium/vilannterol |
A FEV1/FVC ratio of less than 0.7 indicates airflow limitation.
The predicted normal values are dependent on age, gender and height. The tables are provided in Appendix B of 'The Spirometry Handbook'.
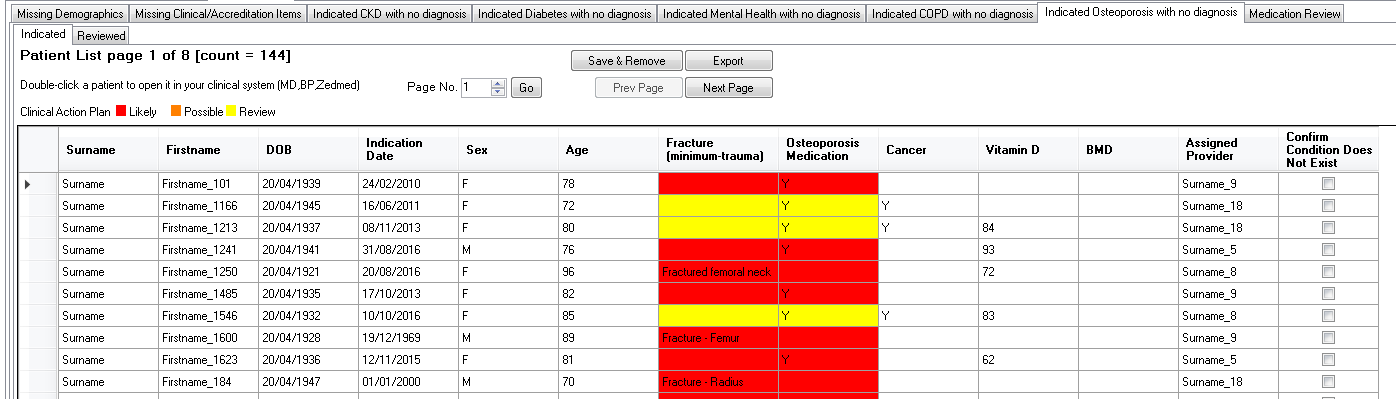
Indicated Osteoporosis with no Diagnosis
The "Indicated Osteoporosis with no diagnosis" prompt will display when a patient meeting the criteria below is opened in the clinical system. The likelihood of the patient having osteoporosis rises from Yellow over Orange to Red as per the table below.
Red - Likely • Aged > 50 with a coded history of a Minimal trauma fracture) • On treatment and without a cancer diagnosis | Orange – Possible • On treatment (Medication) | Yellow – Review • On treatment (Medication) and with a cancer diagnosis |
The report will provide the following fields:
• Surname
• First name
• Date of Birth
• Indication Date
• Sex
• Age
• Minimal Trauma Fracture (Type)
• Treatment (Y/N)
• Cancer (Y/N)
• Vitamin D
• BMD (Date)
• Assigned Provider
The following medications are currently considered:
- Bisphosphonates: alendronate, risedronate, zoledronic acid, pamidronate
- Selective oestrogen modulating drugs: raloxifene
- RANK Ligand Inhibitor: denosumab
- Anabolic drugs: teriparatide/pamathyroid tumor hormone (1-34)
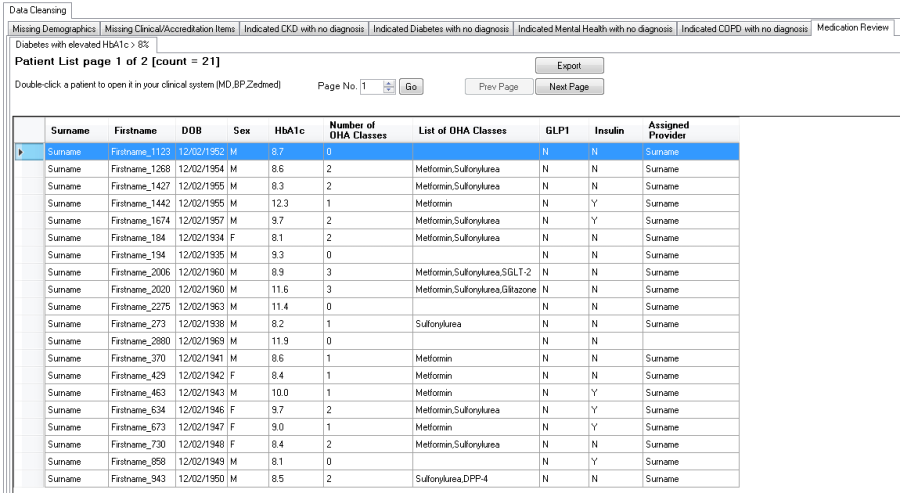
Medication review for Diabetes with elevated HbA1c
The "Medication review > Diabetes with elevated HbA1c" Report will display patients with Type 2 Diabetes whose HbA1c is not being controlled on their current Antidiabetic medications.
Patients will appear on the report where they meet the following criteria:
•Diabetes Type 2
•HbA1c > 8%
The report will display the Antidiabetic medications the patient is currently taking:
Oral Hyperglycaemics | Injectables |
This report provides the opportunity to review and modify the medications being taken to try and bring the HbA1c level into normal range. It also relates to the Diabetes SIP item 'medication review' that should be performed yearly for patients with diabetes.
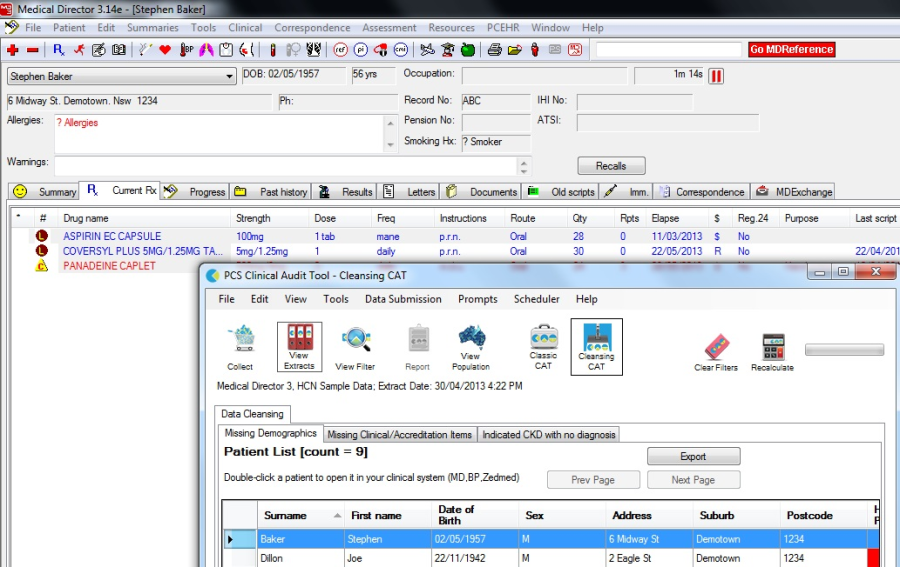
Accessing a Patient Record (All Reports)
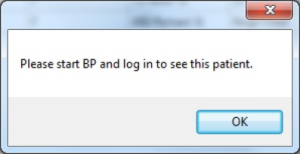
To access a patient record, double-click anywhere on the row containing the patient's details within the displayed report. This will open the patient record within the appropriate clinical system.
Note: The clinical system must be open and logged in for the above step to complete. If the clinical system is not open, an alert message will pop up requesting the clinical system be started.
Clinical systems where this functionality is currently provided are
•Medical Director
•Best Practice
•Zedmed
Other clinical systems are planned to be added.
If your clinical system is not open you will be alerted that you need to start it and login
Condition Does Not Exist Option
CAT4 has an option to remove a patient from the Cleansing View reports by confirming that a particular condition does not exist. This will stop the patient from appearing on the Cleansing View and Cleansing App in Topbar. Only users of Topbar and CAT4 can use this option as it uses the Topbar server to store the information. CAT4 needs to be linked to Topbar in the Edit/Preferences/Topbar settings to activate this function, the details are provided here: Linking CAT4 to Topbar
Excluding patients can be done in CAT4 by using the Cleansing View or in Topbar in the Cleansing App. This guide describes the process for CAT4, please refer to the Cleansing App for Topbar users.
The following reports allow users to exclude patients from future reports and notifications:
- Indicated CKD with no diagnosis
- Indicated Diabetes with no diagnosis
- Indicated Mental Health with no diagnosis
- Indicated COPD with no diagnosis
- Indicated Osteoporosis with no diagnosis
If a patient has new results such as pathology or medications that indicate the condition exists after a patient has been reviewed, the patient will appear again on the 'indicated condition' report. For example:
- A patient taking anti-diabetic medication since many years but the patient doesn't have diabetes and has thus been removed from the 'indicated diabetes without diagnosis' report and the respective Topbar prompt
- This patient has a FBG and/or HbA1c done and the values indicate that the patient might have diabetes, but no diagnosis is recorded.
- Now the patient appears again on the 'indicated diabetes without diagnosis' report and will receive the prompt in Topbar
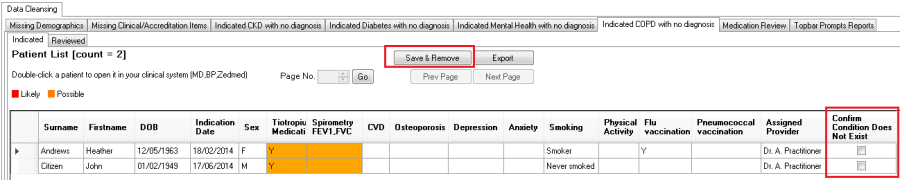
Each report has a column called 'Confirm Condition Does Not Exist' and a 'Save & Remove' button as shown below:
To remove a patient from the list in CAT4 (and the Topbar notifications) for the displayed chronic condition tick the box in the 'Confirm Condition Does Not Exist' and then click the 'Save & Remove' button.
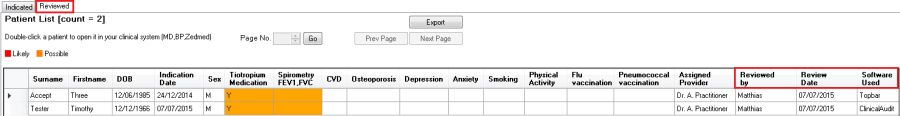
This will immediately remove the patient from the 'Indicated' report tab shown above and add the patient to the second tab called 'Reviewed':
The 'Reviewed' tab shows any patients who have been removed from the 'Indicated' tab for the current indicated chronic condition and also the user name of the person who removed them, the date and software used to remove them. The report will show patients removed by CAT4 as well as those removed by Topbar, as shown above.